Tailored Ground and Excited-State Redox Potentials Through Halogenation and/or Extended π-Conjugation in BODIPY Dye: A Computational Perspective
Ongoing
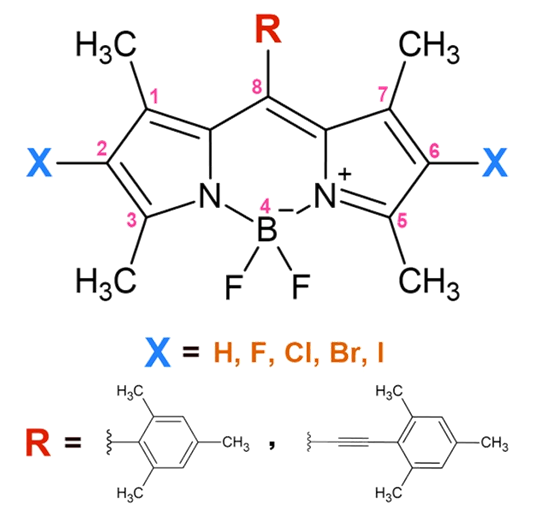
This computational study investigates the effects of halogenation and π-extended conjugation
on the ground-state and excited-state redox potentials of meso-substituted BODIPY using
density functional theory (DFT) and time-dependent DFT (TDDFT). Systematically, we examine
the electronic structure, optical properties, and redox behaviour of ethynyl and non-ethynyl
substituted BODIPYs with halogens (F, Cl, Br, I). Our results reveal that heavy halogenation
stabilizes both singlet and triplet excited states and reduces their energy gaps, while
π-extension via ethynyl group enhances charge transfer and improves the dyes' ability to
accept electrons. These changes suggest large systematic shifts in the redox potentials
and will provide an opportunity to systematically tune BODIPYs for unique performance in
photoredox catalysis. Overall, this research provides insights to design new efficient
metal-free photocatalysts with enhanced electronic properties, advancing sustainable energy
and catalysis.